en
names in breadcrumbs

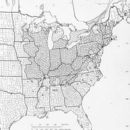
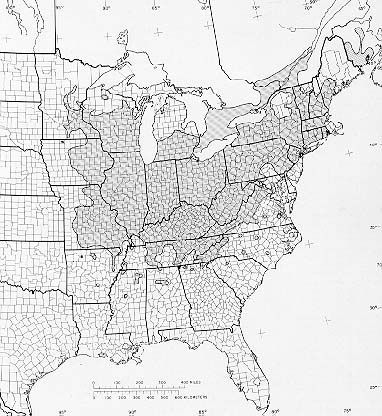
Juglans cinerea (Butternut) is a species of tree in the family Juglandaceae. They have a self-supporting growth form. They are listed as endangered by IUCN and as endangered by COSEWIC. They are associated with freshwater habitat. They are native to Canada, The Contiguous United States, Eastern North America, and United States. They have compound, broad leaves, green flowers, and drupes. Flowers are visited by Myrtle Warbler, Ruby-crowned Kinglet, and White-throated Sparrow. Individuals can grow to 80 feet.
EOL has data for 82 attributes, including: